
The Future Is Here. Built by Gulfstream
At the pinnacle of regulatory evolution, we are developing intelligent, secure AI infrastructure designed to meet the demands of a rapidly changing global landscape. Purpose-built for life sciences, engineered for precision, and grounded in regulatory expertise.
The next era isn’t coming. It’s here.

Unlock the Potential of Gulfstream Intelligence
Our company ethos
The regulatory landscape is evolving. So are we.
At Gulfstream, we’re developing a secure, AI-driven regulatory model built to adapt to shifting global expectations.
Our focus is simple; to increase strategic intelligence, reduce operational budget spend, protect sensitive data, and drive regulatory forward.
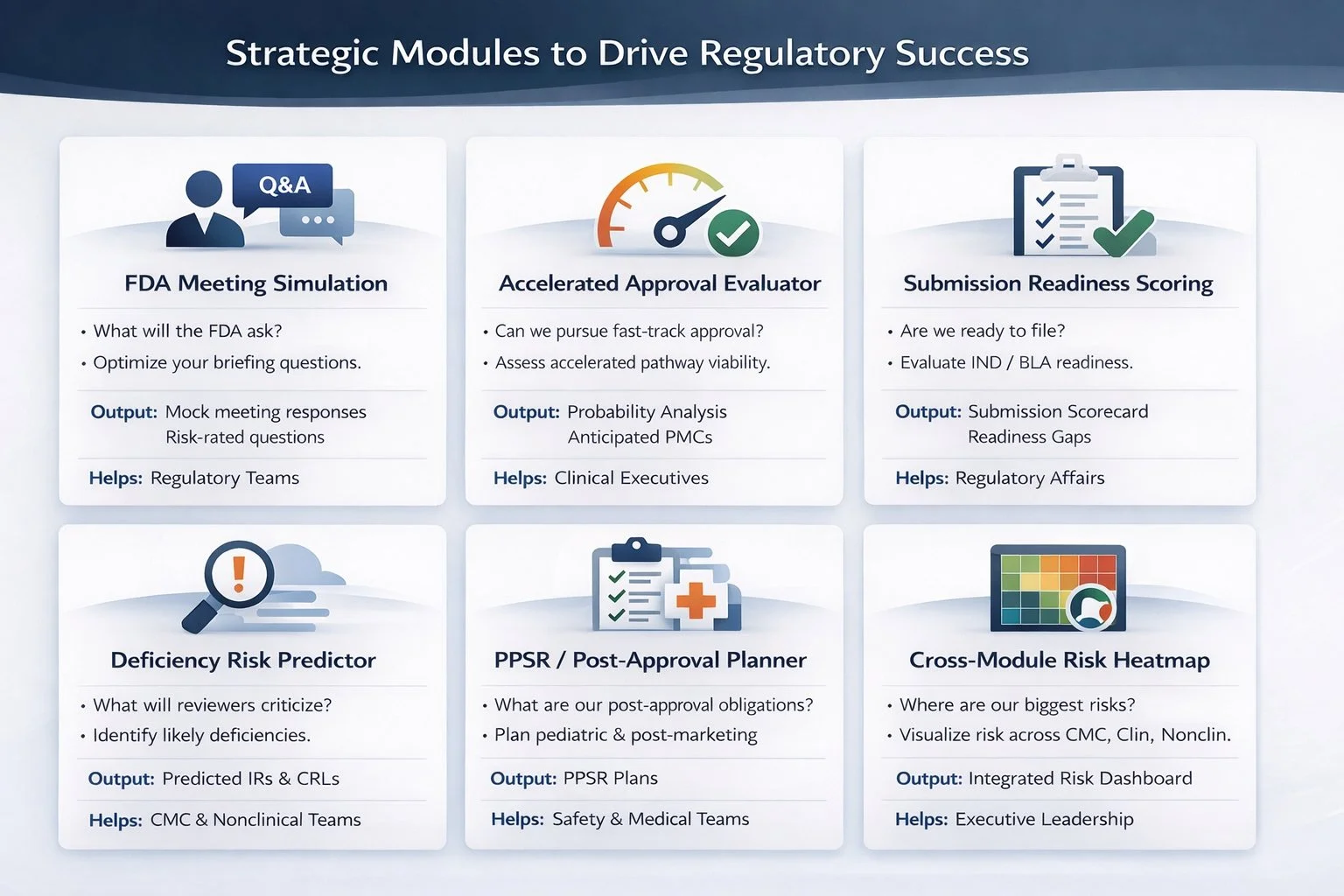
Summary:
Fewer regulatory surprises
Smarter FDA interactions
Defensible decisions
Probability-informed strategy
Executive confidence
Reduce review cycles & IR volume
Avoid unnecessary studies
Decrease likelihood of deficiency letters
Improve meeting outcomes
Align cross-functional strategy early
Cost Effective Tool, Customizable for client’s specific needs.
Follow us on LinkedIn




The Evolution of Regulatory Starts Here
Private Beta Access for Regulatory Leaders (Coming Soon)
Apply for Early Access
“Built for global Regulatory professionals & teams to identify gaps early, reduce risk, and accelerate approvals without compromising data security.”
